Soap dissolution
We all use soap in our daily lives. According to Wikipedia, “Soap is a salt of a fatty acid used in a variety of cleansing and lubricating products.” While using soap, I noticed an interesting phenomenon: a new bar of soap seems to dissolve at much faster rate initially, but as it gets smaller, the rate of dissolution slows down.
At first, this felt like a trivial observation—something we experience daily but never question. Curious, I shared this with a friend (his name is Suyog and he is an Aerospace Engineer), stating Soap dissolves exponentially. Instead of brushing it off, he approached it like an Engineer (or physicist–mathematician) would. His exact reply was: “Interesting!😃”. What followed was a simple yet elegant mathematical explanation that captured this everyday phenomenon well.
I have tried to share our story through this article.
Modeling the Soap
To translate the observation into mathematics, we made a few simplifying assumptions:
The soap is assumed to be a perfect cube.
The soap dissolves uniformly from all exposed surfaces.
The rate of change of volume is proportional to the total surface area of the soap.
Let the side length of the soap cube be \(x\) units.
Geometric Relations
The volume of a cube is given by
\[ V = x^3 \]
The total surface area of the cube is
\[ A = 6x^2 \]
Dissolution Rate Equation
Assuming that the rate at which the soap volume decreases is directly proportional to its surface area:
\[ \frac{dV}{dt} = -k A \]
where:
\(k\) is the dissolution constant, and
the negative sign indicates a decrease in volume over time.
Substituting \(A = 6x^2\) and \(V = x^3\), we write
\[ \frac{dV}{dt} = -6k x^2 \]
Since
\[ V = x^3 \Rightarrow \frac{dV}{dt} = 3x^2 \frac{dx}{dt} \]
we obtain
\[ 3x^2 \frac{dx}{dt} = -6k x^2 \]
Canceling \(x^2\) from both sides gives
\[ \frac{dx}{dt} = -2k \]
Solving the Equation
Integrating with respect to time:
\[ \int dx = -2k \int dt \]
which yields
\[ x = -2kt + C \]
At time \(t = 0\), let the initial volume of the soap be \(V_0\). Then the initial side length is
\[ x_0 = V_0^{1/3} \]
Substituting into the equation, we get
\[ x = V_0^{1/3} - 2kt \]
Final Expression for Soap Volume
Since \(V = x^3\), the volume of the soap at any time \(t\) is
\[ \boxed{V(t) = \left(V_0^{1/3} - 2kt\right)^3} \tag{1} \]
This equation neatly explains the original observation:
When the soap is large, its surface area is high, so it dissolves quickly.
As the soap becomes smaller, the surface area reduces, slowing down the dissolution rate.
The existence of a real root for this equation indicates a finite time at which the soap vanishes completely—confirming the physical validity of the model.
Assuming \(V_0=1\) and \(k=0.05\), plot of equation \((1)\) looks like:
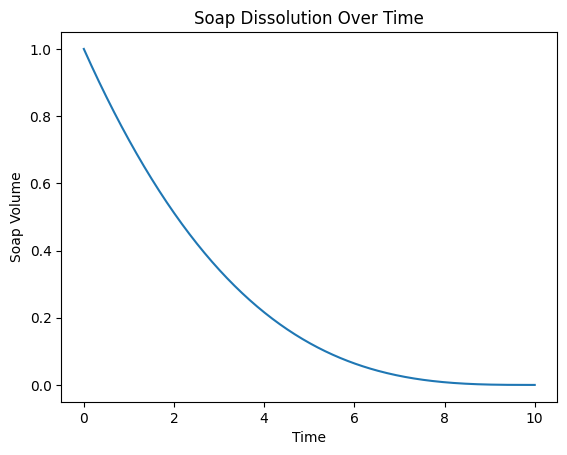
About the Dissolution Constant
The parameter (k) is the dissolution constant of the soap. It depends on several real-world factors, including:
Soap composition
Water temperature
Water hardness
Degree of agitation
The unit of \(k\) is (\(\text{m/s}\)).
Closing Thoughts
What started as a casual observation during a routine activity turned into a satisfying example of how simple mathematics can explain everyday phenomena. It also reinforced an important lesson: even the most mundane experiences can lead to meaningful insights when viewed through a scientific lens.
Sometimes, all it takes is curiosity—and a friend willing to turn that curiosity into equations.